
Back Sellulose Afrikaans Zellulose ALS سليولوز Arabic Celulosa AST Sellüloza Azerbaijani سلولز AZB Целлюлоза Bashkir Цэлюлоза Byelorussian Цэлюлёза BE-X-OLD Целулоза Bulgarian
| |

| |
| Identifiers | |
|---|---|
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.029.692 |
| EC Number |
|
| E number | E460 (thickeners, ...) |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
| (C 12H 20O 10) n | |
| Molar mass | 162.1406 g/mol per glucose unit |
| Appearance | white powder |
| Density | 1.5 g/cm3 |
| Melting point | 260–270 °C; 500–518 °F; 533–543 K Decomposes[2] |
| none | |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
−963,000 kJ/mol[clarification needed] |
Std enthalpy of
combustion (ΔcH⦵298) |
−2828,000 kJ/mol[clarification needed] |
| Hazards | |
| NFPA 704 (fire diamond) | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 15 mg/m3 (total) TWA 5 mg/m3 (resp)[2] |
REL (Recommended)
|
TWA 10 mg/m3 (total) TWA 5 mg/m3 (resp)[2] |
IDLH (Immediate danger)
|
N.D.[2] |
| Related compounds | |
Related compounds
|
Starch |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
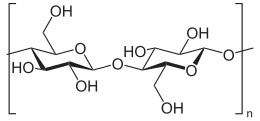
Cellulose is an organic compound with the formula (C
6H
10O
5)
n, a polysaccharide consisting of a linear chain of several hundred to many thousands of β(1→4) linked D-glucose units.[3][4] Cellulose is an important structural component of the primary cell wall of green plants, many forms of algae and the oomycetes. Some species of bacteria secrete it to form biofilms.[5] Cellulose is the most abundant organic polymer on Earth.[6] The cellulose content of cotton fiber is 90%, that of wood is 40–50%, and that of dried hemp is approximately 57%.[7][8][9]
Cellulose is mainly used to produce paperboard and paper. Smaller quantities are converted into a wide variety of derivative products such as cellophane and rayon. Conversion of cellulose from energy crops into biofuels such as cellulosic ethanol is under development as a renewable fuel source. Cellulose for industrial use is mainly obtained from wood pulp and cotton.[6] Cellulose is also greatly affected by direct interaction with several organic liquids.[10]
Some animals, particularly ruminants and termites, can digest cellulose with the help of symbiotic micro-organisms that live in their guts, such as Trichonympha. In human nutrition, cellulose is a non-digestible constituent of insoluble dietary fiber, acting as a hydrophilic bulking agent for feces and potentially aiding in defecation.
- ^ Nishiyama Y, Langan P, Chanzy H (2002). "Crystal Structure and Hydrogen-Bonding System in Cellulose Iβ from Synchrotron X-ray and Neutron Fiber Diffraction". J. Am. Chem. Soc. 124 (31): 9074–9082. doi:10.1021/ja0257319. PMID 12149011.
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0110". National Institute for Occupational Safety and Health (NIOSH).
- ^ Crawford, R. L. (1981). Lignin biodegradation and transformation. New York: John Wiley and Sons. ISBN 978-0-471-05743-7.
- ^ Updegraff D. M. (1969). "Semimicro determination of cellulose in biological materials". Analytical Biochemistry. 32 (3): 420–424. doi:10.1016/S0003-2697(69)80009-6. PMID 5361396.
- ^ Romeo T (2008). Bacterial biofilms. Berlin: Springer. pp. 258–263. ISBN 978-3-540-75418-3.
- ^ a b Klemm D, Heublein, Brigitte, Fink, Hans-Peter, Bohn, Andreas (2005). "Cellulose: Fascinating Biopolymer and Sustainable Raw Material". Angew. Chem. Int. Ed. 44 (22): 3358–3393. doi:10.1002/anie.200460587. PMID 15861454.
- ^ Cellulose. (2008). In Encyclopædia Britannica. Retrieved January 11, 2008, from Encyclopædia Britannica Online.
- ^ Chemical Composition of Wood. Archived October 13, 2018, at the Wayback Machine. ipst.gatech.edu.
- ^ Piotrowski, Stephan and Carus, Michael (May 2011) Multi-criteria evaluation of lignocellulosic niche crops for use in biorefinery processes Archived April 3, 2021, at the Wayback Machine. nova-Institut GmbH, Hürth, Germany.
- ^ Mantanis GI, Young RA, Rowell RM (1995). "Swelling of compressed cellulose fiber webs in organic liquids". Cellulose. 2 (1): 1–22. doi:10.1007/BF00812768. ISSN 0969-0239.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search
